The iStatis Covid-19 Antigen Home Test is developed for use in everyday settings to help you feel confident in your status so that you can take action to reduce exposure and prevent transmission.
Features:
- Non-Invasive: With a non-invasive test, we make knowing your COVID-19 statis easy by removing the discomfort of rapid testing.
- Quality: iStatis is made by bioLytical, a trusted, experienced test manufacturer based in Canada. Our facility is ISO 13485:2016 MDSAP certified, so you can be sure your test works to keep you safe.
- Trusted Results: One of the best tools against COVID-19 is testing, and with the iStatis COVID-19 Antigen Home Test’s high accuracy, you can be confident in your test results, helping you make informed decisions so you can get back to your life safely.
- Easy-to-Use: Rapid tests should simplify taking care of your health. With clear instructions, an easy-to-use platform, and a simple and painless procedure, we make knowing your status simple and fast so that you can get back to your life with confidence.
Product Information:
Quantity: 5 tests/pack
Sample type: Shallow Nasal Swab
Storage temperature: 2–30°C (do not freeze)
Test time: 15 minutes (do not read results after 30 minutes)
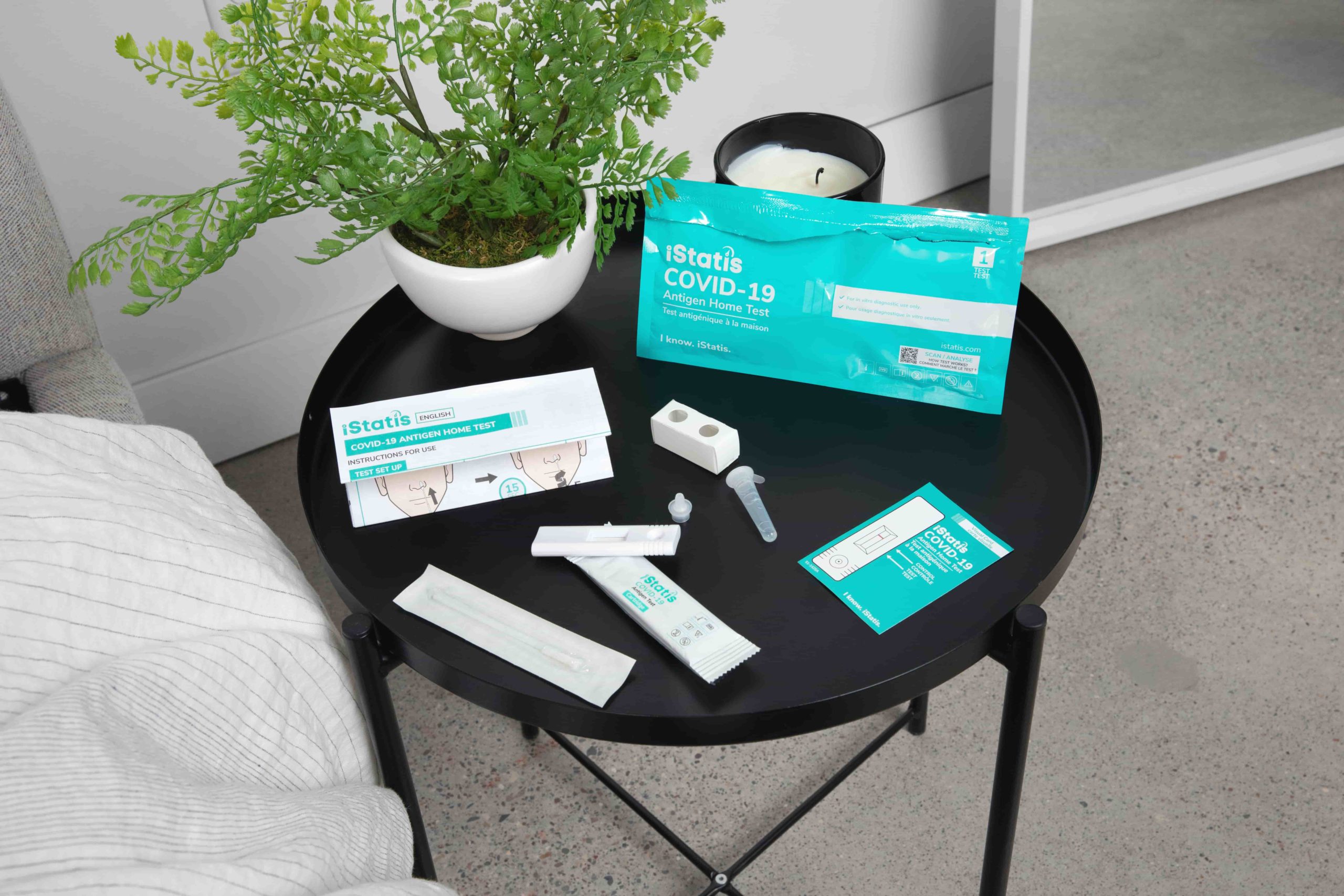
In the Box
5 Test Cartridges
5 Nasal Swabs
5 Buffer Vials and Buffer Vial Caps
1 Vial Holders
1 Package Inserts
1 Visual Cards
Intended Use
The iStatis COVID-19 Antigen Test is a single use, visually read, lateral-flow in vitro qualitative immunoassay intended for the detection of nucleocapsid protein antigen from SARS-CoV-2 in anterior nasal swab specimens collected from individuals 18 years or older who are suspected of COVID-19 by their healthcare provider within the first seven days of symptom onset, or from individuals without symptoms or other epidemiological reasons to suspect COVID-19 when tested twice over two or three days with at least 24 hours and no more than 48 hours between tests. iStatis COVID-19 Antigen Test is suitable for near-patient or point-of-care (POC) testing use only.
Results are for the identification of the SARS-CoV-2 nucleocapsid protein antigen. The antigen is generally detectable in anterior nasal swab specimens during the acute phase of infection. Positive results indicate the presence of nucleocapsid protein, but the clinical correlation with patient history and other diagnostic information is necessary to determine infection status. Positive results do not preclude bacterial infection or co-infection with other viruses. The agent detected may not be the definite cause of disease.
Negative results do not preclude SARS-CoV-2 infection and should not be used as a primary source of data for treatment or patient management, including infection control. If acute infection is suspected, direct testing for SARS-CoV-2 is necessary.
Results should instead be considered within the context of a patient’s recent exposures, history, and the presence of symptoms consistent with COVID-19, and confirmed with a molecular assay, if necessary, for any further decisions.
For serial testing programs, additional confirmatory testing with a molecular test for negative results may be necessary, if there is a high likelihood of SARS-CoV-2 infection, such as an individual with a close contact with COVID-19 or with suspected exposure to COVID-19 or in communities with high prevalence of infection. Additional confirmatory testing with a molecular test for positive results may also be necessary, if there is a low likelihood of SARS-CoV-2 infection, such as in individuals without known exposures to SARS-CoV-2 or residing in communities with low prevalence of infection.
The iStatis COVID-19 Antigen Test is intended for use by medical professionals or operators trained in performing tests in point of care settings.
By purchasing this product, you agree to follow the manufacturer’s intended use and the sale conditions of this website.





























































Natalie Rush (verified customer) –
I keep a stash of the iStatis tests at home. They’re perfect for last-minute checks before gatherings or travel.
Kimberly Jackson (verified customer) –
In a world full of uncertainties, iStatis is something I can count on. Accurate results, every time.
Breanna Byrd (verified customer) –
Being a frequent traveler, the iStatis test has become an essential for me. It’s compact, easy, and reliable.
Tricia Gibson (verified customer) –
I’ve tried several antigen tests, and the iStatis COVID-19 test is by far the most reliable. Results come quickly and are easy to understand.
William Grant (verified customer) –
Super pleased with my purchase from Rapid Test & Trace. The iStatis COVID-19 Antigen Test is easy to use, and the results are rapid as promised!