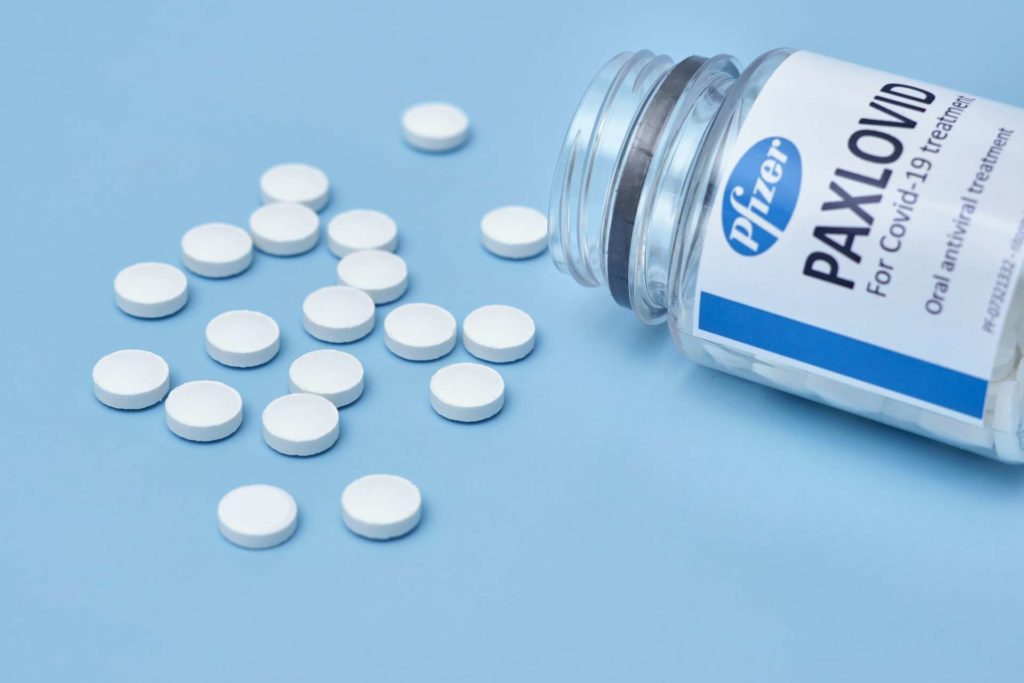
Paxlovid is the most effective and trusted COVID-19 antiviral medication currently available in Canada. It was issued an Emergency Use Authorization and was approved by Health Canada for prescription to patients in January of 2022.
WHAT IS PAXLOVID?
Paxlovid is an antiviral drug developed by Pfizer specifically to help a treated patient’s immune system fight off COVID-19 infection, avoid developing symptoms that lead to severe illness and prevent hospitalizations as a result.
It is meant to stop the continued spread of the infection within the body, which will effectively help to prevent symptomatic progression in patients to the point of hospitalization.
Paxlovid consists of two medicinal ingredients, nirmatrelvir and ritonavir. They work together to interrupt the COVID-19 replication process within our cells, which directly contributes to the moderation of symptoms.
NIRMATRELVIR & RITONAVIR
Paxlovid has been alternatively described as “ritonavir-boosted nirmatrelvir”.
This is because ritonavir actually does not act directly on the covid-19 virus itself, but instead serves to “boost” the effect of nirmatrelvir in the body.
Nirmatrelvir
Nirmatrelvir is the primary active ingredient in the Paxlovid antiviral medication.
It is a protease inhibitor molecule that works to suppress the COVID-19 virus and essentially halt its lifecycle.
Protease inhibitors are compounds that interfere with the ability of specific enzymes to break down proteins and perform their function.
Nirmatrelvir specifically works by inhibiting a cysteine residue in the 3CL protease of the COVID-19 virus.
Without the proper functioning of the 3CL protein enzyme, the virus cannot replicate itself and as a result will not be able to continue spreading within the host.
Ritonavir
Ritonavir is also a protease inhibitor that acts as an antiretroviral medication that has been used alongside other medications to treat HIV/AIDS.
Ritonavir is included in low-doses in Paxlovid COVID-19 treatments. It serves to decrease the rate at which nirmatrelvir is broken down by the body.
This characteristic increases the half-life of nirmatrelvir, which means that the compound will persist in treated patients for a longer period of time.
As a result, ritonavir effectively increases the concentration of nirmatrelvir, and subsequently optimizes treatment effectiveness.
WHO SHOULD TAKE PAXLOVID?
Paxlovid has been approved by Health Canada for individuals who have tested positive for COVID-19, are experiencing mild to moderate symptoms, and are at high risk for the development of serious illness.
Speak with your health care provider if you are unsure of your current health status and whether or not are considered high risk.
Eligible patients will meet a set of criteria:
- Must have tested positive for COVID-19 (by way of PCR test or rapid antigen test)
- 12 years of age or older – weighing at least 40 kg (88 lbs.)
- Mild/moderate symptoms of COVID-19
- No known or suspected kidney impairments
- No known or suspected liver impairments
- Do not require hospitalization due to severe symptoms at the time of treatment request
- Symptom onset within five days of treatment request
- One or more risk factors for progression to severe symptoms of COVID-19
Risk factors/conditions include:
- Over 70 years of age
- Over 60 years of age with less than three vaccine doses
- Obesity
- Diabetes
- Heart disease, hypertension, cognitive heart failure
- Chronic respiratory disease (cystic fibrosis etc..)
- Cerebral palsy
- Sickle cell disease
- Pregnancy
Health care providers can provide more insight as to whether or not you have serious underlying conditions or risk factors that would make you.
HOW TO GET A PAXLOVID PRESCRIPTION IN CANADA
Paxlovid is Canada’s first oral antiviral treatment for mild to moderate COVID-19. It was authorized by Health Canada on January 17th of 2022.
Provinces and territories have been given their own authority to determine who can prescribe and dispense Paxlovid to patients.
In Saskatchewan, Alberta, Quebec and Newfoundland and Labrador, pharmacists are authorized to prescribe Paxlovid to patients who have been tested and can show proof of a positive result.
In other provinces, like Ontario and British Columbia, Paxlovid can only be prescribed by a doctor or nurse practitioner.
Getting a Paxlovid prescription through RT Medical is a simple process that can be done from the comfort of your own home.
We provide a streamlined telehealth service that connects you with one of our highly qualified health care providers.
After a quick and easy virtual assessment, a Paxlovid prescription will be provided for eligible patients to fill at their local community pharmacies.
Get ahead of COVID-19 with Paxlovid.
AUTHORIZATION OF TREATMENT IN CANADA
The Public Health Agency of Canada only authorizes treatments of any kind after a thorough scientific review of the safety, efficacy and quality of the components involved.
Authorization may come with a set of conditions that the manufacturer must meet to help ensure that the quality and effectiveness of the products is consistent.
Companies must also have a Health Canada approved risk management plan (RMP) when applying for treatment authorization.
Risk Management Plans Include:
- The treatment’s safety profile
- Information on how risks will be minimized or prevented
- Concrete plans for further studies and discovery about the safety and effectiveness of the treatment
- Information regarding the monitoring and assessing of measures that minimize the potential treatment risks
PAXLOVID & KIDNEY DISEASE
Health Canada Treatment with Paxlovid is not recommended for patients who have a history of severe kidney disease.
Patients who have been diagnosed with moderate cases of kidney disease can still safely take Paxlovid, however their dosage should be adjusted accordingly to ensure safety.
Dosage
Normal renal function/mild renal impairment
Regular dosage – 300 mg nirmatrelvir & 100 mg ritonavir taken together twice daily for 5 days.
Moderate renal impairment
Adjusted dosage – 150 mg nirmatrelvir & 100 mg ritonavir taken together twice daily for 5 days.
Severe renal impairment
Paxlovid treatment is not recommended.
ANTIVIRAL TREATMENTS IN CANADA
Health Canada has authorized a number of COVID-19 antiviral treatments since the pandemic began.
These treatments are available for all provinces and territories to implement in their own respective health care systems. Public health measures specific to each location will determine the appropriate course of action for administering these drugs to eligible patients.
Other treatments that have been authorized by Health Canada for COVID-19 include:
- Remdesivir (Veklury)
- Bamlanivimab
- Casirivimab and imdevimab
- Sotrovimab
- Nirmatrelvir and ritonavir (Paxlovid)
- Tixagevimab and cilgavimab (Evusheld)
PAXLOVID ANTIVIRAL PILL EFFECTIVENESS
Pfizer provided clinical trial data collected by studies conducted from July to December of 2021 that indicated the relative effectiveness of Paxlovid treatments.
Published data suggests that treatment of symptomatic COVID-19 patients who are at higher risk for progression to severe disease resulted in an 89% risk reduction.
Researchers have also since concluded that the use of Paxlovid in individuals who are fully vaccinated remains effective in comparison to those who are not fully vaccinated.
Paxlovid is the leading antiviral treatment for COVID-19 in Canada. Other antiviral treatments that have been authorized by Health Canada are simply not as effective both from a logistical and medical standpoint.
FAQ
Is Paxlovid a Replacement for Vaccines?
Paxlovid treatment is NOT a replacement for COVID-19 vaccines.
Vaccines are administered in order to promote health and prevent infection in the first place, while antiviral pills like Paxlovid are meant to manage symptoms after infection has occurred.
Are Antiviral Treatments Safe For Pregnancy?
Oral antiviral treatments (pill form) for COVID-19 have been recommended for use in individuals who are pregnant or breastfeeding by the National Institutes of Health and the American College of Obstetricians.
Regardless, you should always consult with your health care provider to assess your specific situation and health needs before proceeding with COVID-19 treatments.
What is Included in Paxlovid COVID-19 Treatments?
Paxlovid treatments consist of two different medicines provided in pill form.
Each daily dosage card contains 4 pink nirmatrelvir pills, as well as 2 white ritonavir pills. These 6 pills make up two daily doses to be taken over 5 days, for a total of 10 doses.
Each dose consists of 3 pills – 2 nirmatrelvir and 1 ritonavir taken at the same time or in immediate succession.
If you are looking for more information, or would like to book a consultation to get a Paxlovid prescription, visit us at www.getpaxlovid.ca.