Description
Artron COVID-19 Antigen Home Test is a rapid lateral flow immunoassay intended for the qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in self-collected anterior nasal (nares) swab samples obtained from individuals aged 14 years or older or adults collected nasal swab specimens from individuals aged two years or older. This test is intended for individuals suspected of COVID-19 within the first seven days of symptom onset when tested at least twice over three days with at least 48 hours between tests, and for individuals without symptoms or other epidemiological reasons to suspect COVID-19 when tested at least three times over five days with at least 48 hours between tests.
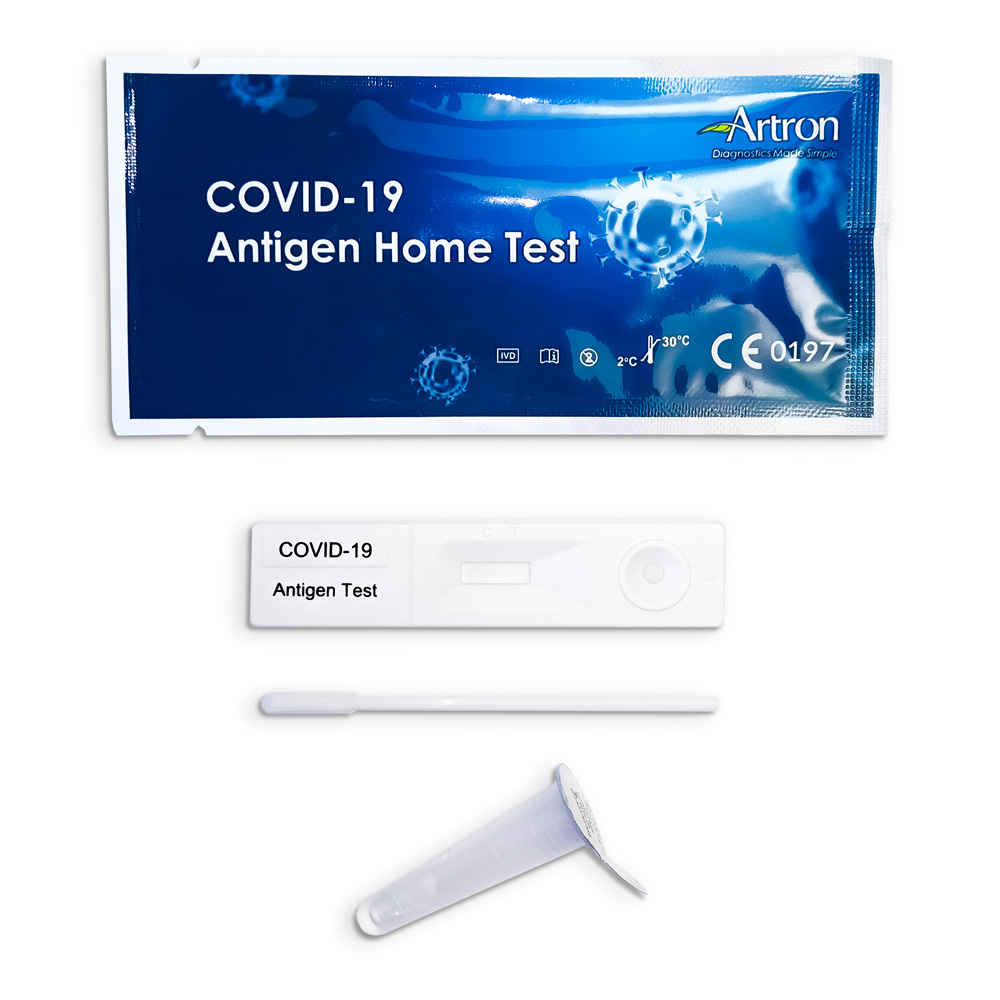
What’s included
- 25 Test cassettes with desiccant in individual pouch
- 25 Sterilized nasal swabs
- 25 Extraction tubes
- 25 Extraction tube caps
- 1 Instructions for use (IFU)
- 1 Quick reference guide
Materials required but not provided:
Instructions
How to use
- Tear off the aluminum foil seal from the extraction tube.
- Remove a nasal swab at the stick end from the pouch.
- Gently insert the SWAB 1⁄2 – 3⁄4 inch (1.3-1.9 cm) into the nostril, depending on the size of the person’s nose. Firmly rub the SWAB in a circular motion around the inside wall of EACH NOSTRIL at least 5 times or more for at least 15 seconds.
Be sure to rub BOTH nostrils with the SAME SWAB.
NOTE: If you are swabbing others, please wear a face mask.
With children, you may not need to insert the swab as far into the nostril.
For very young children, you may need another person to steady the child’s head while swabbing.
NOTE: Failure to swab properly may cause false negative results.
- Immediately insert the swab into the extraction tube. Swirl the swab tip vigorously in the buffer fluid at least 10 times.
- Remove the swab by rotating against the extraction tube while squeezing the sides of the tube to release the liquid from the swab. Dispose of the swab in the trash.
- Close the extraction tube with the provided extraction tube cap and push firmly onto the tube.
Test procedures
- Get the test cassette from the sealed pouch by tearing it at the notch and placing the cassette on a flat and dry surface. Do not touch the test window.
- Hold the extraction tube vertically (not at an angle) above the sample well, and slowly add 4 drops of the specimen without air bubbles into the sample well. DO NOT touch the card with the dropper tip while dispensing.
- Read and interpret the test result in 15-30 minutes. The test result should not be read and interpreted after 30 minutes.
- All used test components should be disposed of in the household waste.
Warnings and precautions
- For in vitro diagnostic use only.
- The test Is designed only for the detection of nasal swab specimens.
- This test is only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens.
- All kit components are single use items. Do not use with multiple specimens. Do not reuse the test cassette.
- Do not use if the pouch seal or the packaging is compromised.
- Do not use after the expiry date shown on the pouch.
- Do not mix and interchange different specimens.
- The swabs in the kit are approved for use with Artron COVID-19 Antigen home test. Do not use other swabs.
- Do not touch swab tip when handling the swab specimen.
- If the test is stored refrigerated, ensure that the test units are brought to room temperature (15-30°C) at least 30 mins before performing testing.
- Use the test device immediately after opening the pouch.
- INVALID RESULTS can occur when an insufficient drop of sample is added to the test cassette. To ensure delivery of adequate volume, hold vial vertically, ½ inch above the swab well, and add drops slowly.
- Wash hands thoroughly before and after finishing the testing.
- Dispose of kit components and patient samples in household trash.
- Keep out of children’s reach.
- If the extraction buffer contacts the skin or eye, flush with copious amounts of water.
Intended Use
Artron COVID-19 Antigen Home Test is a rapid lateral flow immunoassay intended for the qualitative detection of SARS-CoV-2 nucleocapsid protein from SARS-CoV-2. This test is for home use with self-collected anterior nasal (nares) swab specimens from individuals aged 14 years or older or adults and who are suspected of COVID-19 within seven days of symptom onset or epidemiological criteria or adult collected nasal swab specimens from individuals aged 4 years or older who are suspected of COVID-19 within the first seven days of symptom onset or epidemiological criteria. The rapid test device is for home use only and is intended to be used as an aid I the diagnosis of SARS-CoV-2 infection.
FAQs
Is the Artron Covid-19 Home Test Accurate?
The Artron COVID-19 Rapid Test is designed for high accuracy, utilizing advanced technology to detect the presence of the virus. However, like all rapid tests, there may be a small margin of error.
Can Children Use the Artron Covid-19 Rapid Test?
Yes, the Artron COVID-19 Rapid Test is suitable for children under adult supervision. It’s important to follow the instructions carefully for accurate results.
How Should I Interpret the Test Results?
The test results are indicated by the presence of lines on the testing device. We have provided the instructions to help you correctly interpret the results.
What Should I Do If I Test Positive For COVID-19 with the Artron Home Test?
If you test positive, it’s important to follow local health guidelines, which may include self-isolation and contacting a healthcare provider for further advice and testing.
Can I Use The Artron Covid-19 Home Test To Travel Internationally?
The acceptability of the Artron COVID-19 Home Test for international travel depends on the specific requirements of the destination country. Always check the latest travel guidelines before using this test for travel purposes.
























































Tiffany Roberts –
The Artron Antigen COVID-19 Rapid Test exceeded my expectations. It was a convenient and reliable option for early diagnosis of SARS-CoV-2 infection. The test had a high sensitivity and specificity, which gave me confidence in its accuracy.
Allen Dougherty –
I had a great experience with the Artron Antigen COVID-19 Rapid Test. It was easy to use and provided me with reliable results. I would highly recommend this test for professional and laboratory use.
Shannon Jensen –
I travel a lot for work, and the Artron Antigen COVID-19 Rapid Test has become a staple in my bag. It’s easy to use and gives results in no time. A lifesaver for those on the go.
Andrea Nichols –
I recently used the Artron Antigen COVID-19 Rapid Test and I couldn’t be happier with the results. The test was very convenient to use and provided a quick screening result. The package included everything I needed, from the test cassettes to the swabs and mixing tubes. Overall, a great product for professional use
Christian Anderson –
Needed a quick test before a family gathering, and Artron Antigen COVID-19 Rapid Test delivered. Got my result in 15 minutes. It’s a great tool, but always be sure to test regularly and maintain safety protocols.